Iron »
PDB 2grz-2hk6 »
2grz »
Iron in PDB 2grz: 5NS Photoproduct of the M37V Mutant of Scapharca Hbi
Protein crystallography data
The structure of 5NS Photoproduct of the M37V Mutant of Scapharca Hbi, PDB code: 2grz
was solved by
J.E.Knapp,
R.Pahl,
V.Srajer,
W.E.Royer Jr.,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 16.85 / 1.60 |
| Space group | C 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 93.040, 43.930, 83.440, 90.00, 121.95, 90.00 |
| R / Rfree (%) | 10.6 / 10.9 |
Iron Binding Sites:
The binding sites of Iron atom in the 5NS Photoproduct of the M37V Mutant of Scapharca Hbi
(pdb code 2grz). This binding sites where shown within
5.0 Angstroms radius around Iron atom.
In total 2 binding sites of Iron where determined in the 5NS Photoproduct of the M37V Mutant of Scapharca Hbi, PDB code: 2grz:
Jump to Iron binding site number: 1; 2;
In total 2 binding sites of Iron where determined in the 5NS Photoproduct of the M37V Mutant of Scapharca Hbi, PDB code: 2grz:
Jump to Iron binding site number: 1; 2;
Iron binding site 1 out of 2 in 2grz
Go back to
Iron binding site 1 out
of 2 in the 5NS Photoproduct of the M37V Mutant of Scapharca Hbi
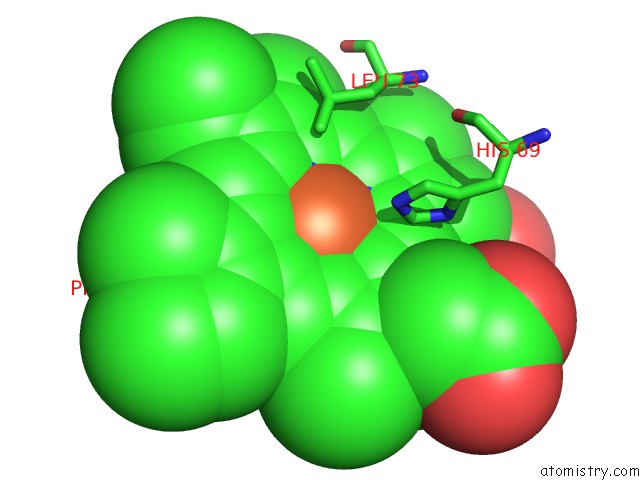
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 1 of 5NS Photoproduct of the M37V Mutant of Scapharca Hbi within 5.0Å range:
|
Iron binding site 2 out of 2 in 2grz
Go back to
Iron binding site 2 out
of 2 in the 5NS Photoproduct of the M37V Mutant of Scapharca Hbi
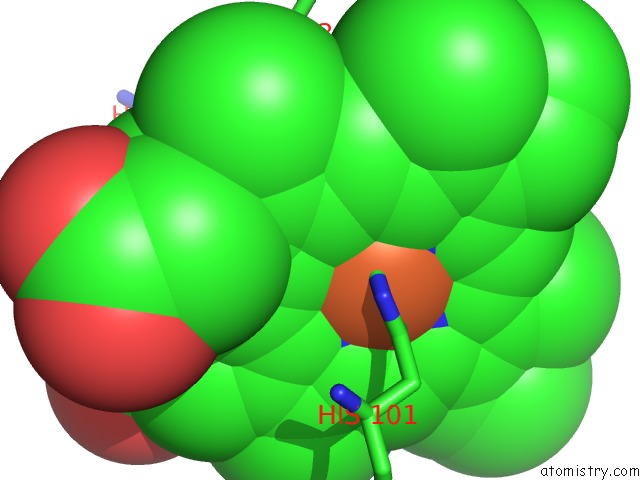
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 2 of 5NS Photoproduct of the M37V Mutant of Scapharca Hbi within 5.0Å range:
|
Reference:
J.E.Knapp,
R.Pahl,
V.Srajer,
W.E.Royer Jr..
Allosteric Action in Real Time: Time-Resolved Crystallographic Studies of A Cooperative Dimeric Hemoglobin. Proc.Natl.Acad.Sci.Usa V. 103 7649 2006.
ISSN: ISSN 0027-8424
PubMed: 16684887
DOI: 10.1073/PNAS.0509411103
Page generated: Sat Aug 3 22:48:56 2024
ISSN: ISSN 0027-8424
PubMed: 16684887
DOI: 10.1073/PNAS.0509411103
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW