Iron »
PDB 1sdl-1stq »
1sqx »
Iron in PDB 1sqx: Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
Enzymatic activity of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
All present enzymatic activity of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A:
1.10.2.2;
1.10.2.2;
Protein crystallography data
The structure of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A, PDB code: 1sqx
was solved by
L.Esser,
B.Quinn,
Y.F.Li,
M.Zhang,
M.Elberry,
L.Yu,
C.A.Yu,
D.Xia,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 40.00 / 2.60 |
| Space group | I 41 2 2 |
| Cell size a, b, c (Å), α, β, γ (°) | 154.385, 154.385, 590.271, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 23.3 / 28.1 |
Iron Binding Sites:
The binding sites of Iron atom in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
(pdb code 1sqx). This binding sites where shown within
5.0 Angstroms radius around Iron atom.
In total 5 binding sites of Iron where determined in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A, PDB code: 1sqx:
Jump to Iron binding site number: 1; 2; 3; 4; 5;
In total 5 binding sites of Iron where determined in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A, PDB code: 1sqx:
Jump to Iron binding site number: 1; 2; 3; 4; 5;
Iron binding site 1 out of 5 in 1sqx
Go back to
Iron binding site 1 out
of 5 in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
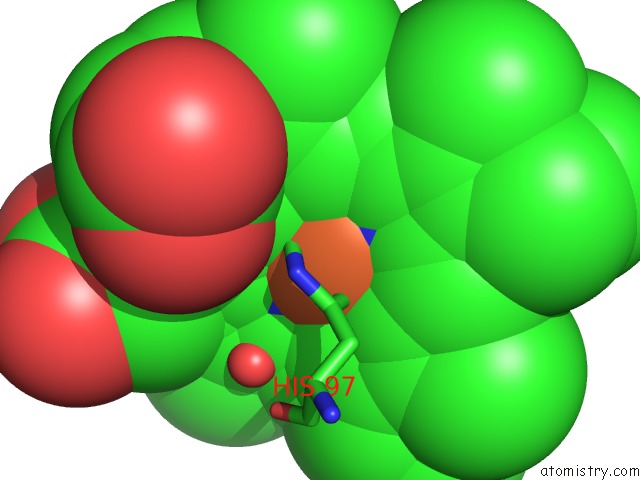
Mono view
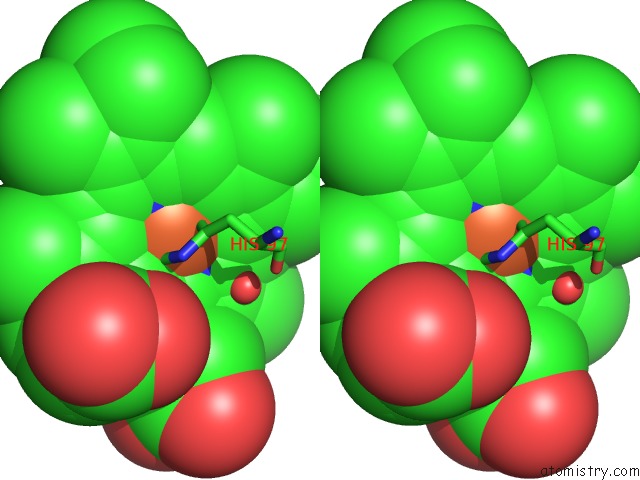
Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 1 of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A within 5.0Å range:
|
Iron binding site 2 out of 5 in 1sqx
Go back to
Iron binding site 2 out
of 5 in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
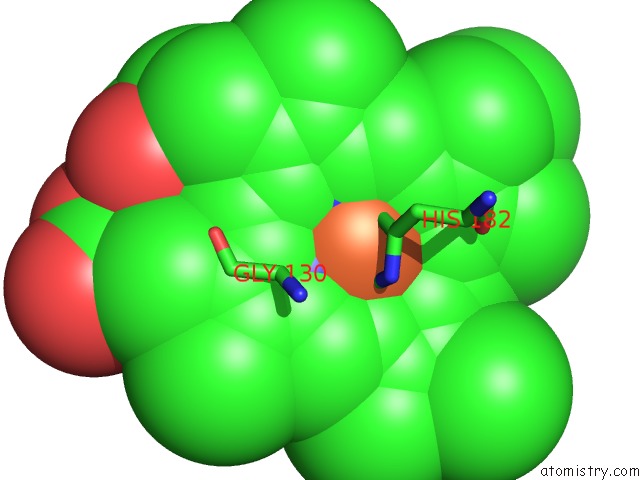
Mono view
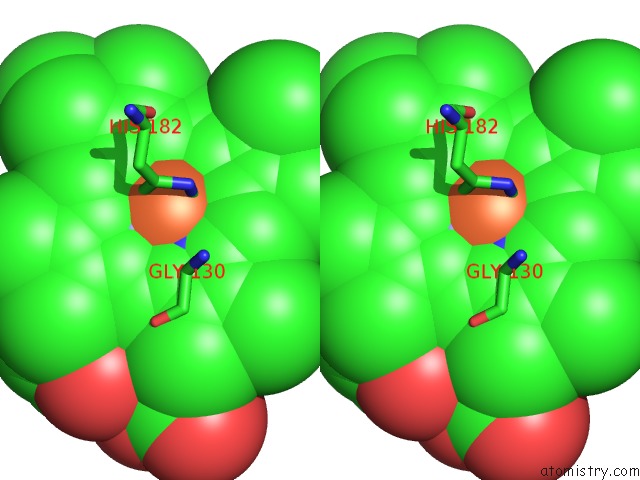
Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 2 of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A within 5.0Å range:
|
Iron binding site 3 out of 5 in 1sqx
Go back to
Iron binding site 3 out
of 5 in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
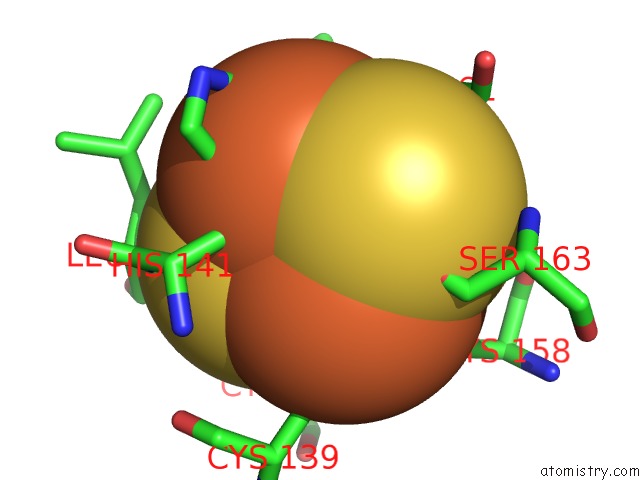
Mono view
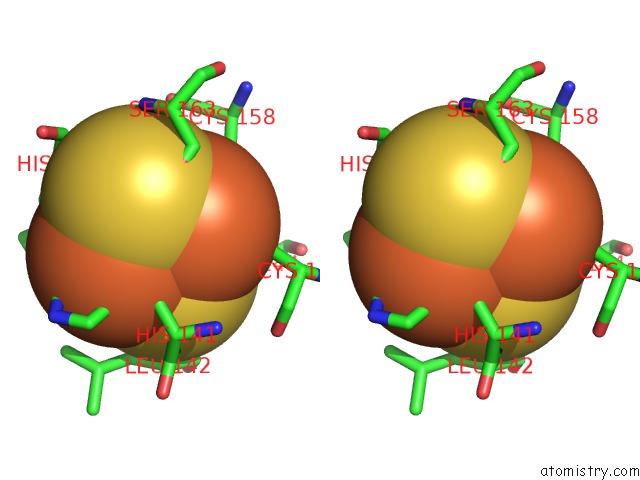
Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 3 of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A within 5.0Å range:
|
Iron binding site 4 out of 5 in 1sqx
Go back to
Iron binding site 4 out
of 5 in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
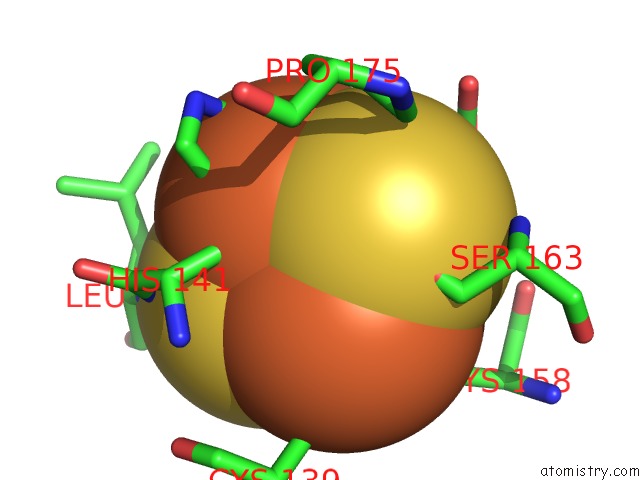
Mono view
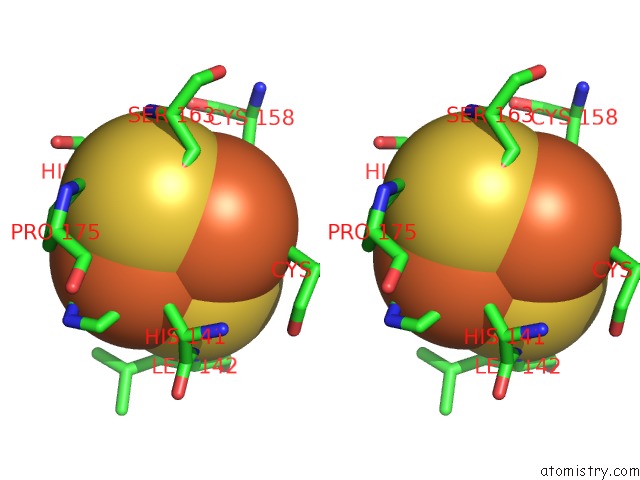
Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 4 of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A within 5.0Å range:
|
Iron binding site 5 out of 5 in 1sqx
Go back to
Iron binding site 5 out
of 5 in the Crystal Structure Analysis of Bovine BC1 with Stigmatellin A
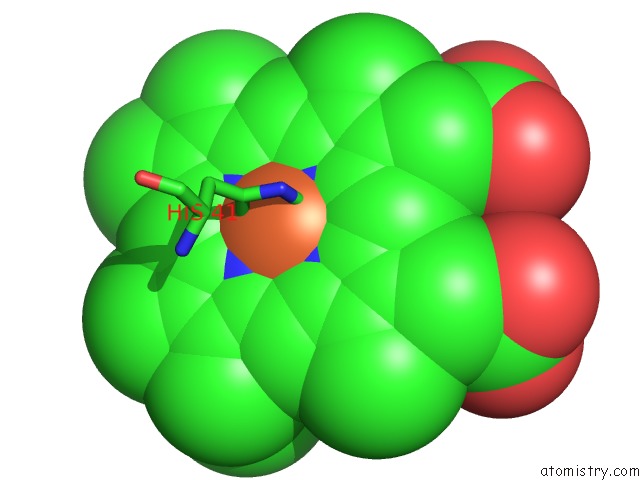
Mono view
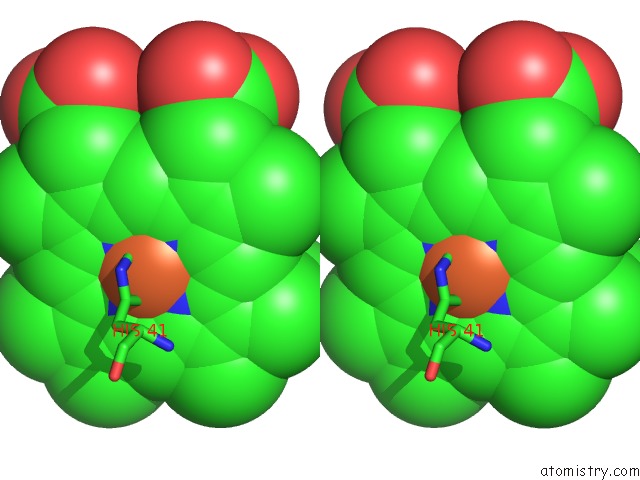
Stereo pair view

Mono view

Stereo pair view
A full contact list of Iron with other atoms in the Fe binding
site number 5 of Crystal Structure Analysis of Bovine BC1 with Stigmatellin A within 5.0Å range:
|
Reference:
L.Esser,
B.Quinn,
Y.F.Li,
M.Zhang,
M.Elberry,
L.Yu,
C.A.Yu,
D.Xia.
Crystallographic Studies of Quinol Oxidation Site Inhibitors: A Modified Classification of Inhibitors For the Cytochrome Bc(1) Complex. J.Mol.Biol. V. 341 281 2004.
ISSN: ISSN 0022-2836
PubMed: 15312779
DOI: 10.1016/J.JMB.2004.05.065
Page generated: Sat Aug 3 14:51:21 2024
ISSN: ISSN 0022-2836
PubMed: 15312779
DOI: 10.1016/J.JMB.2004.05.065
Last articles
Cl in 3ZZWCl in 3ZXV
Cl in 3ZXR
Cl in 3ZZE
Cl in 3ZXQ
Cl in 3ZX6
Cl in 3ZXD
Cl in 3ZX2
Cl in 3ZX0
Cl in 3ZWX